Primary liver cancers develop within a complex microenvironment where tumor cells closely interact with immune cells, stromal cells, and the extracellular matrix. This microenvironment is strongly influenced by the digestive tract, the main reservoir of the gut microbiota, whose bacterial derivatives transit to the liver via the gut–liver axis. These bacteria and their metabolites can modulate the tumor immune phenotype, influencing local immunity, tumor tolerance, and response to therapies.
Our team studies the tumor microenvironment and its role in immune tolerance in primary liver cancers, particularly hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (ICC). Leveraging our expertise, we analyze interactions between tumor cells, the immune system, and the microbial ecosystem to identify new therapeutic levers. We explore the influence of the gut microbiota, bacterial products, and certain innate immunity effectors on tumor physiology and immunity. The aim is to identify microbial biomarkers and develop innovative strategies to optimize responses to immunotherapies.
Our research is organized around three complementary axes:
(1) Deciphering the mechanisms of hepatic and loco-regional immune tolerance and uncovering their connection with the human microbiota.
(2) Demonstrating the ability of specific microbes or microbial products to overcome tumor-induced immunosuppression in preclinical models.
(3) Investigating the impact of the innate immunity effector REG3A on organ homeostasis and liver carcinogenesis.
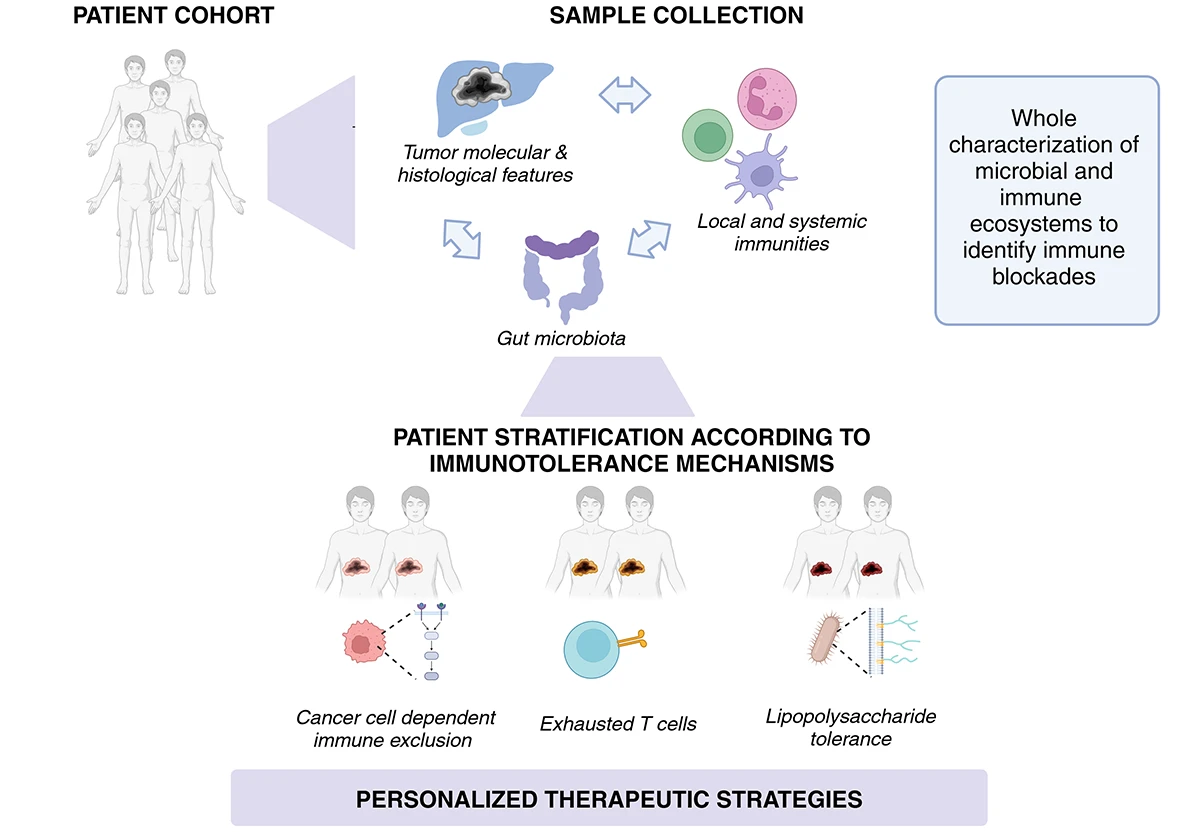
Tumor immune tolerance is a major barrier to the effectiveness of immunotherapies in primary liver cancers. Using an integrative approach combining immunological, transcriptomic, and metabolomic analyses, our team was among the first to identify and characterize the immune phenotypes and underlying mechanisms of immune tolerance present in ICC and HCC tumors (Job et al., 2020; Shalhoub et al., 2024).
Our current research aims to unravel the complex interactions between the tumor microenvironment, local and systemic immunity, and the gut microbiota, in order to better understand the pathophysiology of these cancers and identify new therapeutic targets.
Leveraging patient cohorts followed at the Hepatobiliary Center (CHB), we focus on three main objectives:
This work enables the identification of metagenomic and immunological biomarkers predictive of immunotherapy response and paves the way for personalized medicine approaches, which will be further validated in preclinical models in Axis 2.
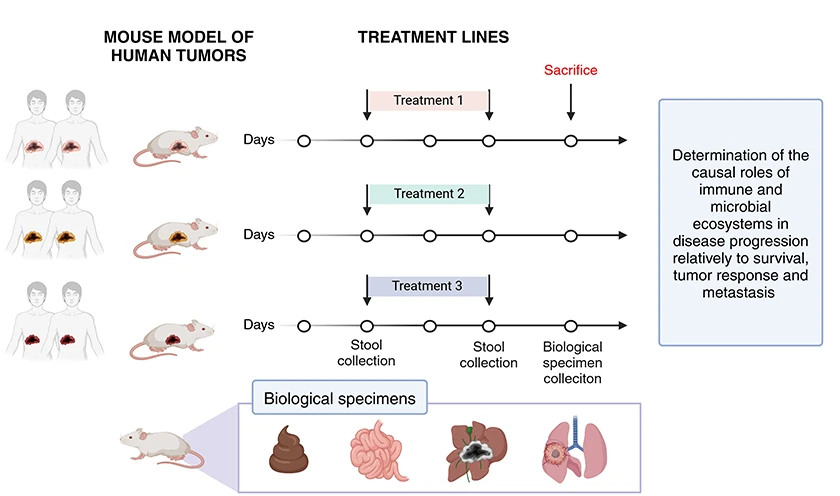
The gut microbiota directly influences the progression of primary liver cancers, notably via the portal circulation and the immune interface. While its role is well established in chronic liver diseases such as metabolic steatohepatitis (MASH), its impact on liver carcinogenesis remains insufficiently characterized.
Our goal is to explore the potential of microbial regulators to reshape the immunosuppressive tumor microenvironment into an immunoactive phenotype capable of slowing tumor progression. Our work has already highlighted the central role of REG3A, an antimicrobial and antioxidant peptide from innate immunity, in controlling microbial diversity and the production of immunomodulatory metabolites. These properties position REG3A as a promising therapeutic lever to overcome tumor immune tolerance and restore treatment efficacy (Moniaux et al., 2024; Darnaud et al., 2018).
Within this framework, we aim to experimentally demonstrate the causal role of the intestinal microbiota and its derivatives in:
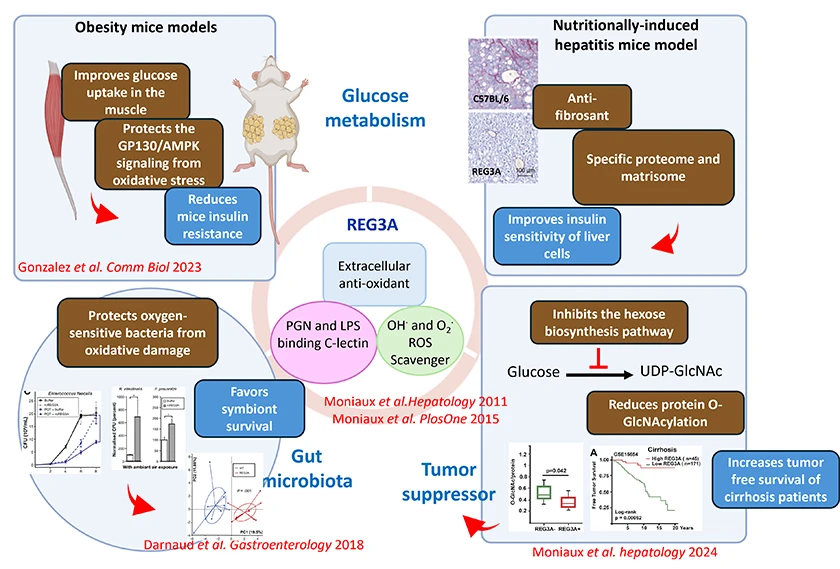
REG3A is a C-type lectin and innate immunity effector with antioxidant properties that allows it to trap reactive oxygen species (ROS). It plays a central role at the interface between the gut microbiota and organ homeostasis, acting both on the host and its commensal flora. Our research has highlighted several key functions of REG3A:
Perspectives
Our future work aims to unravel the mechanisms through which REG3A modulates the interactions between the microbiota, metabolome, and hepatic immune ecosystem, in the context of nutrition-related metabolic disease and primary liver cancers. We will focus on two main directions:
These studies rely on an integrative, translational approach, combining metagenomic, transcriptomic, metabolomic, and immunological analyses, in close collaboration with clinicians and biotechnology companies specializing in immunotherapy.
Ultimately, this research could lead to the development of combined therapeutic strategies that integrate microbiota modulation and immunotherapy, with the potential to significantly improve the management of patients with hepatocellular carcinoma (HCC) or intrahepatic cholangiocarcinoma (ICC).
High-throughput Data

Imaging
Animal Models

Molecular and Cellular Biology



















